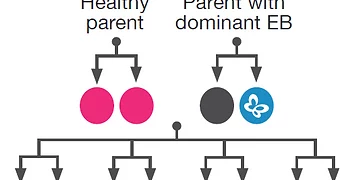
The U.S. Congress steadily keeps up its never-ending task of crafting new, better laws. At the same time, biotechnology businesses and DNA testing services struggle through the expensive and challenging process of persuading government regulators to approve new goods.
According to congressional databases that track federal bills, the Senate and House members on both sides of the aisle introduced a staggering 982 regulatory proposals in the 106th Congress alone, which began January 26. These proposals mention the subjects of biotechnology, genetic engineering, the human genome, genetically modified plants and animals, cloning, use of face recognition app and international trade of genetically engineered products.
Biotech Within A Broader Framework
Most of these laws mention biotech within a broader framework, such as patent reform, commerce in agricultural products, or healthcare. Even while the billion-dollar DNA testing business and genetic engineering business currently only produces around 100 commercially accessible agriculture and medicine products, the sector’s involvement in many legislative proposals demonstrates that biotechnology is profoundly ingrained in society.
Recent federal laws span a wide range of initiatives to liberate either further or further restrict biotech, most of which will either shrivel from neglect or die a lingering death between an obscure subcommittee and a floor vote. Sen. Richard Lugar’s (R-IN) S 566 would amend the Agricultural Trade Act of 1978 to exempt “value-added” products—the polite term for genetically modified medicines and chemicals—from financial penalties in current and future international trade agreements.
Biotech and Medicare and Medicaid
In a regulatory twist, however, biotech lobbyists claim that the reform of Medicare and Medicaid is this year’s most hotly debated and possibly combustible legislative topic. According to Nancy Bradish Meyers, director of government relations for the Biotechnology Industry Organization (BIO), one of the most significant trade associations representing domestic and foreign biotech firms, “our overarching concern in recent years has been the increasing number of bills that indirectly or tangentially affect the field of biotechnology.” “At this moment, an increasing number of members believe in biotechnology, but we have to make sure they are aware of how laws that look unrelated to the business might have a detrimental impact.”
Lowering the Cost of Drugs
More than 200 pieces of legislation have been introduced in the House and Senate due to efforts to wholly or partly repair Medicare, the most recent regulatory poster child for Congress. The ideas to lower the cost of drugs are the ones that Bradish Meyers is most concerned about. Sen. Tim Johnson (D-SD) and Sen. Edward Kennedy (D-MA), two influential senators in the field of healthcare, introduced S. 731 on March 25. This bill will significantly lower the cost of prescription medications for Medicare beneficiaries. Rep. Thomas Allen is the sponsor of H.R. 664, the bill’s counterpart in the House (D-ME).
In the Medicare/drug price dispute, the primary concern of biotech proponents is that decreased drug costs may endanger biotech research and development and result in a “medicine deficit.” Added Bradish Meyers: “Investors in startups are always considering the payoffs. The possibility of pricing limits may deter investment in biotech businesses as they now rely on private funding for their research.”
Lawmakers and Lobbyists and DNA Testing
Bills to preserve medical records are a matter of equal importance. Lawmakers have proposed three measures in the Senate, including Sen. Robert Bennet’s “The Medical Information Protection Act of 1999.” (R-UT). The proposed legislation would ensure patient privacy while yet allowing for research. Strong patient advocate lobbyists are fervently advocating for comprehensive medical record privacy. Bradish Meyers claims that biotech firms need access to medical information and DNA testing to finish the human clinical studies required to release medicine for sale.
Center for Technology Assessment
Old-school biotech opponents have changed tactics as biotech businesses bargain with patient groups. They occupy state and federal courts rather than seeking fights on Capitol Hill. According to Andrew Kimbrell, executive director of the International Center for Technology Assessment (CTA), biotechnology regulation has been delegated to organizations where already-existing laws that Congress has previously amended are applied to control paternity testing and genetically engineered products. Federal regulators are attempting to fit the square pegs of medicine and pesticide rules into the round holes of biotech, which Kimbrell calls “a weird aberration.”
Low-Level Presence
Adventitious presence often referred to as low-level presence (LLP), is the accidental and unintentional admixture of small quantities of one kind of seed, grain, or food product with another. Adventitious presence (A.P.) is a fact of plant biology, the manufacturing of sources, and the distribution of commodity crops that cannot be avoided. Pollen flow, volunteering, mixing during harvesting, transport, storage, processing, human mistake, and accidents are just a few of the variables that might result in accidental presence.
Agricultural Biotechnology Stewardship Technical Committee
The Agricultural Biotechnology Stewardship Technical Committee (ABSTC) is a group of agricultural biotechnology firms and technology suppliers that are Bt corn registrants and collaborate to solve scientific problems essential to properly stewarding plant biotechnology products.